Iodine, an essential trace element in human health and various industrial applications, possesses intriguing properties that spark curiosity about its conductive behavior. Electrical conductivity, a fundamental concept in materials science and chemistry, determines a substance’s ability to conduct electric current.
Likewise, thermal conductivity governs how efficiently heat is transferred through a material. The question of whether iodine is conductive leads us to explore its diverse physical and chemical properties, from its molecular structure to its behavior in different states.
In this article, we delve into the conductive nature of iodine, examining its electrical and thermal conductivity under various conditions. We unravel the mysteries surrounding the conductivity of iodine in its solid, liquid, and dissolved forms, shedding light on its role as both an insulator and conductor.
From its significance in human health, particularly in thyroid function, to its applications in the chemical industry, photography, and as an antiseptic, understanding the conductive properties of iodine unveils its multifaceted nature. So, join us on this scientific journey as we unravel the conductivity of iodine and its implications in our world.
What is Iodine?
Iodine is an important element for human health and is an essential micronutrient that is required for the synthesis of thyroid hormones, which are responsible for regulating metabolic processes in the body. These hormones are responsible for maintaining normal body temperature, heart rate, and blood pressure, as well as helping to ensure proper growth and development.

Iodine deficiency is a serious problem worldwide, particularly in countries where soil and water are poor sources of iodine. It can result in the development of goiter, which is an enlargement of the thyroid gland. In severe cases, iodine deficiency can also lead to cretinism, a condition characterized by severe mental retardation and stunted growth in children. Fortunately, iodine deficiency is largely preventable through dietary measures, such as consuming iodized salt and eating foods that are rich in iodine, such as seaweed, fish, and dairy products.
Aside from its importance for human health, iodine has a variety of other uses. It is commonly used in medical settings as a disinfectant, due to its ability to kill bacteria, viruses, and fungi. It is also used in the production of a wide range of industrial chemicals, such as polymers, plastics, and dyes. Iodine is also commonly used in photography as a component of film emulsions.
Iodine has gained significant attention in recent years as a potential propellant for electric propulsion systems used in spacecraft. This is due to iodine’s high energy density, low toxicity, and relatively low cost compared to other propulsion options.
In 2021, an in-orbit demonstration of an iodine-electric propulsion system was successfully carried out. The propulsion system, which used solid iodine pellets as a propellant, demonstrated high performance and efficiency. The system was also found to be highly compatible with spacecraft architecture and had simple logistics requirements.
In addition, the use of iodine in electric propulsion systems is attractive from an environmental standpoint. Unlike traditional chemical rocket engines, which rely on the combustion of propellants that produce a significant amount of pollution, iodine-based electric propulsion systems emit no harmful pollutants into space.
Properties of Iodine:
Physical Properties of Iodine:
- Appearance and State: At room temperature, iodine presents itself as a shiny, dark purple-black solid. It has a characteristic lustrous appearance that distinguishes it from other elements. Interestingly, iodine sublimes, meaning it transitions directly from a solid to a violet-colored vapor without passing through the liquid state when heated;
- Odor and Taste: Iodine has a distinct, pungent odor that can be easily recognized. It also imparts a slightly metallic taste when ingested in small amounts;
- Solubility: Iodine exhibits limited solubility in water, which means it doesn’t dissolve readily in this liquid. However, it dissolves well in certain organic solvents like ethanol and diethyl ether, making it useful in various laboratory processes;
- Density: Iodine has a relatively high density compared to many other elements, with a value of about 4.93 grams per cubic centimeter;
- Melting and Boiling Points: The melting point of iodine is approximately 113.7 degrees Celsius (236.7 degrees Fahrenheit), while its boiling point is around 184.3 degrees Celsius (363.7 degrees Fahrenheit);
- Crystalline Structure: In its solid state, iodine forms crystals with a layered structure, held together by weak Van der Waals forces between the layers. This property contributes to its characteristic shiny appearance;

Chemical Properties of Iodine:
- Reactivity: Iodine is a moderately reactive element. It is less reactive than other halogens like chlorine and bromine but more reactive than elements in the noble gas group. In the presence of heat or light, iodine can undergo certain chemical reactions;
- Oxidation States: Iodine can exhibit various oxidation states, ranging from -1 to +7, but its most common and stable oxidation states are -1, +1, +3, +5, and +7. The -1 oxidation state is observed in compounds called iodides, while the +1 state is found in compounds known as iodates;
- Halogen Characteristics: As a halogen, iodine shares certain characteristic properties with its group members, including high electronegativity, which means it has a strong attraction for electrons when forming chemical bonds;
- Displacement Reactions: Iodine can displace less reactive halogens from their compounds in displacement reactions. For instance, it can displace chlorine from sodium chloride to form sodium iodide;
- Reaction with Starch: One of the unique chemical properties of iodine is its reaction with starch, resulting in a dark blue or black color. This reaction is often used to test for the presence of starch in various substances [2];
Compounds of Iodine:
Iodine forms various compounds due to its reactivity and ability to combine with other elements. Some of the common compounds of iodine include:
- Iodides: Iodides are compounds where iodine exists in the -1 oxidation state. Potassium iodide (KI) and sodium iodide (NaI) are examples of iodides used in medicine, photography, and analytical chemistry;
- Iodates: Iodates contain iodine in the +5 oxidation state. An example of iodate is potassium iodate (KIO3), which is used as a source of iodine in iodized salt and as an oxidizing agent in chemical reactions;
- Hydrogen Iodide: Hydrogen iodide (HI) is a colorless gas formed by the direct combination of hydrogen and iodine. It is used in organic synthesis and as a reducing agent in certain chemical reactions;
- Iodine Pentoxide: Iodine pentoxide (I2O5) is a powerful oxidizing agent and dehydrating agent used in organic synthesis and the preparation of certain iodine compounds;
- Organic Iodine Compounds: Various organic compounds contain iodine, and they are commonly used in medicine as disinfectants and antiseptics;
Occurrence and Abundance of Iodine
Iodine is a relatively rare element in the Earth’s crust, but it is crucial for various biological processes and has a notable presence in certain environments. Its distribution and abundance are influenced by geological, chemical, and biological factors.

Geological Sources:
- Seawater: The most significant reservoir of iodine on Earth is the world’s oceans. Iodine exists in seawater primarily in the form of iodide ions (I^-). The concentration of iodine in seawater is relatively low, typically around 0.06 parts per million (ppm). Despite its low concentration, the vastness of the oceans results in a substantial global iodine reservoir;
- Marine Organisms: Marine organisms, particularly seaweeds and certain types of algae, have the ability to accumulate iodine from seawater. They concentrate iodine in their tissues, making them rich sources of this element. Additionally, various marine animals, such as fish and shellfish, also contain iodine, reflecting their iodine intake through their diet;
- Soil and Rocks: Iodine can be found in soil and rocks, but its abundance in terrestrial environments is generally low. The iodine content in soil is influenced by factors such as weathering of rocks, volcanic activity, and the presence of iodine-rich minerals;
- Iodine-Rich Soils: In some regions, particularly near coastal areas, certain soils may have higher iodine content due to the deposition of marine aerosols and sediments carried by the wind;
Abundance:
Iodine is considered a trace element, meaning it occurs in relatively low concentrations compared to major elements like oxygen, silicon, and aluminum. The average abundance of iodine in the Earth’s crust is estimated to be around 0.45 parts per million (ppm), making it one of the less abundant elements.
Despite its low overall abundance, iodine plays a critical role in various biological processes, particularly in the human body. It is an essential micronutrient required for the synthesis of thyroid hormones, which are crucial for regulating metabolism and promoting proper growth and development.
Importance in Human Health:
Iodine deficiency is a significant global health issue, affecting millions of people in various regions around the world. Insufficient iodine intake can lead to thyroid disorders, such as goiter (enlarged thyroid gland), hypothyroidism (underactive thyroid), and developmental impairments, especially in children.
To combat iodine deficiency disorders (IDD), many countries have implemented iodine fortification programs. One of the most successful initiatives has been the iodization of table salt, where small amounts of potassium iodide or potassium iodate are added to table salt to ensure adequate iodine intake in the population.
Uses of Iodine
1. Thyroid Health
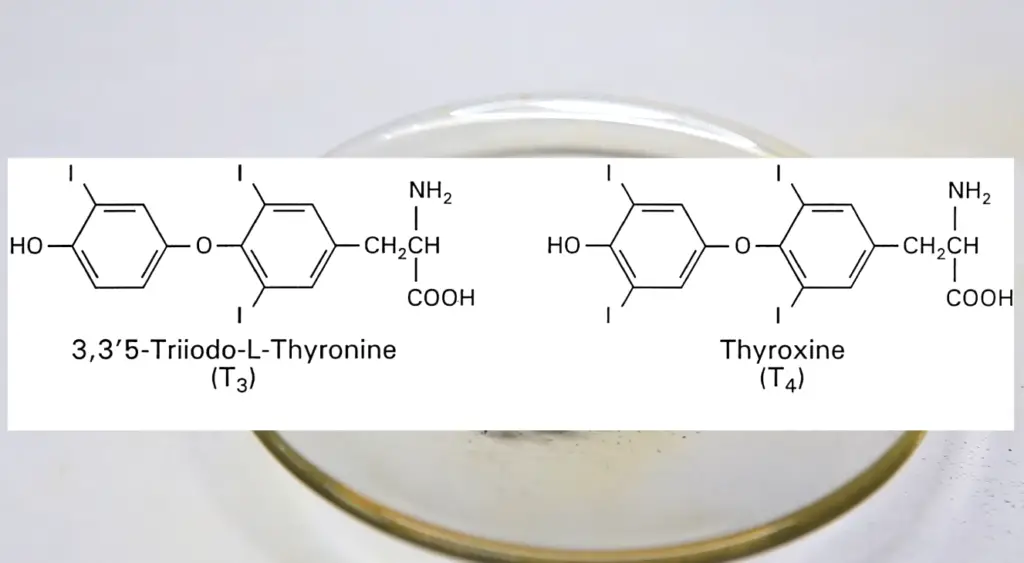
One of the most critical uses of iodine is in maintaining proper thyroid function. The thyroid gland utilizes iodine to produce essential hormones like thyroxine (T4) and triiodothyronine (T3). These hormones are crucial for regulating metabolism, energy production, and the development of the nervous system. Adequate iodine intake is vital for preventing thyroid disorders, such as goiter and hypothyroidism, and for ensuring optimal cognitive and physical development, especially during pregnancy.
2. Iodized Salt
Iodine deficiency is a significant global health issue, affecting millions of people in various regions. To combat this problem, many countries have implemented iodine fortification programs, where small amounts of iodine are added to table salt. Iodized salt ensures a steady and accessible source of iodine for populations, helping to prevent iodine deficiency disorders (IDD) and promoting better overall health.
3. Disinfectants and Antiseptics
Iodine’s antiseptic properties make it valuable in medical and healthcare settings. Povidone-iodine, a widely used iodine-based antiseptic, is used to clean and disinfect skin before surgical procedures, injections, and wound care. It effectively kills bacteria, viruses, and fungi, reducing the risk of infections and promoting faster healing.
4. Pharmaceuticals
Iodine and iodine compounds are employed in the pharmaceutical industry for the production of various medications. Iodine’s antimicrobial properties make it an essential component in the formulation of antibiotics, antifungal drugs, and antiviral agents. Additionally, iodine is used to prepare contrast agents for medical imaging procedures like X-rays and CT scans.
5. Water Purification
In emergencies or situations where access to clean water is limited, iodine can be used as a water disinfectant. Iodine tablets or solutions can be added to untreated water to kill harmful microorganisms, making it safer for drinking and other purposes.
6. Veterinary Applications
Iodine finds applications in veterinary medicine as well. It is used in the treatment of various animal diseases, disinfecting wounds, and preventing infections in livestock and companion animals.

7. Chemical Industry
Iodine and its compounds are utilized in the chemical industry for various purposes. For example, iodine pentoxide (I2O5) is a potent oxidizing agent used in organic synthesis. Iodine is also employed in the production of certain dyes and stains, particularly in the textile industry.
8. Photography
In traditional photography, iodine is used as part of the process to develop photographic films. The light-sensitive silver halide crystals in the film react with iodine, contributing to the formation of the final image.
Industrial and Commercial Applications
Apart from its importance in human health, iodine, and its compounds find applications in various industries and commercial products. Some notable uses include:
- Pharmaceuticals: Iodine and iodine compounds are used in the production of medications and antiseptics. Povidone-iodine, for example, is a widely used antiseptic in the medical field;
- Photography: Iodine is used in traditional photography as part of the process to develop photographic films;
- Dyes and Stains: Iodine is employed in the manufacturing of certain dyes and stains, particularly in the textile industry;
- Water Treatment: Iodine is sometimes used as a disinfectant for water treatment, especially in emergency situations;
Is Iodine a Conductor of Electricity?
In fact, iodine is an insulator, meaning it does not allow the flow of electric current through it.
In its solid state, iodine forms a crystalline structure composed of iodine molecules held together by weak Van der Waals forces between the layers. These forces do not allow the movement of electrons, which is essential for the conduction of electricity. As a result, solid iodine does not conduct electricity and is considered an electrical insulator.

However, it is important to note that iodine can become a conductor under certain conditions. When iodine is dissolved in certain solvents like water or ethanol, it can form ions and conduct electricity. In this state, iodine exists as iodide ions (I^-), which are responsible for carrying the electric charge [3].
Why is Iodine a Bad Conductor of Electricity?
Iodine is a bad conductor of electricity due to its molecular structure and its inability to provide free electrons necessary for the conduction of electric current.
In its solid state, iodine exists as a diatomic molecule (I2), where two iodine atoms are covalently bonded together. The electronegativity of iodine is relatively high, which means the two atoms in the I2 molecule have a strong attraction for their shared electrons. As a result, the electrons are not free to move within the molecule, and there are no mobile charge carriers that can carry an electric current. This lack of free electrons makes solid iodine an electrical insulator.
Another reason why iodine is a bad conductor of electricity is its crystal lattice structure in the solid state. In the crystal lattice, iodine molecules are held together by weak Van der Waals forces between the layers. While these forces give solid iodine its characteristic shiny appearance, they also prevent the movement of electrons between the layers, further hindering its ability to conduct electricity.
To become a conductor of electricity, a material must have mobile charge carriers that can freely move in response to an electric field. Metals, for example, are good conductors of electricity because they have a “sea” of delocalized electrons that are not strongly bound to specific atoms and can move freely throughout the material.

In contrast, iodine lacks this “sea” of mobile charge carriers, which is why it is a bad conductor of electricity in its solid state. However, as mentioned earlier, when iodine is dissolved in certain solvents or in its gaseous state, it can become a conductor due to the formation of ions and the presence of free electrons that can move and carry an electric charge.
Is Iodine Magnetic or Non-magnetic?
Iodine is considered nonmagnetic in its natural form. It does not exhibit any significant magnetic properties under normal conditions.
In its solid state, iodine exists as a diatomic molecule (I2), where two iodine atoms are covalently bonded together. The electronic configuration of iodine does not lead to the presence of unpaired electrons, which are necessary for a substance to exhibit magnetic behavior.
Magnetic properties in materials arise from the alignment and interaction of electrons within their atomic or molecular structures. If a substance has unpaired electrons, it may be magnetic or exhibit paramagnetic behavior (weak attraction to a magnetic field). However, iodine lacks unpaired electrons in its ground state, and as a result, it does not display magnetic properties.
It is important to note that some compounds of iodine, such as organometallic complexes or coordination compounds containing transition metals, may exhibit magnetic behavior if the metal ions involved have unpaired electrons. However, elemental iodine itself, in its pure form, is nonmagnetic.
Does Iodine Conduct Heat?
Iodine is a relatively poor conductor of heat. While it can conduct heat to some extent, it does so at a slower rate compared to many other materials, particularly metals.
The ability of a material to conduct heat is determined by its thermal conductivity, which measures how efficiently heat is transferred through the substance. Metals, with their delocalized electrons that can easily transfer thermal energy, typically have high thermal conductivity. On the other hand, nonmetals like iodine usually have lower thermal conductivity due to their atomic or molecular structures, which are less conducive to efficient heat transfer.

While iodine is not an efficient conductor of heat, its properties can be utilized in specific applications. For example, iodine is sometimes used as a component in certain heat transfer fluids due to its relatively low thermal conductivity. In these applications, it serves as a “thermal buffer” to help regulate and control heat transfer [4].
How Does the Conductivity of Iodine Change Between Solid and Liquid?
Solid State Conductivity
In its solid state, iodine exists as a diatomic molecule (I2), with two iodine atoms covalently bonded together. The electrons in the I2 molecule are localized and tightly bound to the atoms, resulting in a lack of free or mobile charge carriers. As a consequence, solid iodine is considered a nonconductor of electricity. The absence of free electrons also leads to low thermal conductivity, making solid iodine a relatively poor conductor of heat.
Additionally, in its solid form, iodine molecules are arranged in a crystalline structure, with weak Van der Waals forces holding the layers together. This arrangement further impedes the transfer of heat and electrical charges between the iodine molecules, contributing to its insulating properties.
Liquid State Conductivity
As iodine is heated and reaches its melting point of around 113.7 degrees Celsius (236.7 degrees Fahrenheit), it undergoes a phase transition from a solid to a liquid state. In this liquid state, the iodine molecules gain enough energy to break free from their fixed positions in the crystal lattice, allowing them to move more freely.
The transition to the liquid state introduces a new dynamic environment where the iodine molecules are now mobile, and their interactions change. While still not an excellent conductor of electricity, liquid iodine shows increased electrical conductivity compared to its solid form. This is because the increased molecular mobility allows for some ionic dissociation, leading to the presence of mobile ions (I^-) that can carry electric charge to a limited extent.
The thermal conductivity of liquid iodine also improves relative to its solid state. With increased molecular mobility and weaker intermolecular forces, heat transfer becomes more efficient in the liquid phase. However, it is still considerably lower than the thermal conductivity observed in most metals and other highly conductive materials.
FAQ:
1. What is electrical conductivity in chemistry?
Electrical conductivity in chemistry refers to the ability of a substance to conduct electric current. It is a measure of how easily electric charges, usually carried by ions or electrons, can move through the material. Conductors have high electrical conductivity as they allow electric charges to flow freely, while insulators have low electrical conductivity and hinder the flow of electric charges.
2. What kind of charge does iodine have in water?
In water, iodine primarily exists as iodide ions (I^-). These ions have a negative charge, as they have gained an extra electron, leading to an overall negative charge.
3. Which is the best type of insulator for iodine?
The best type of insulator for iodine would be materials that can prevent the transfer of heat or electricity efficiently. Given iodine’s weak electrical and thermal conductivity, most solid materials can act as insulators for iodine, including plastics, ceramics, and nonmetals.
4. Is iodine a conductor or insulator?
Iodine is a poor conductor of both electricity and heat in its solid form due to its molecular structure, and it is considered an insulator. However, in its liquid state, iodine exhibits some degree of electrical conductivity due to the presence of mobile ions, although it is still not an efficient conductor.
5. Does iodine attract electrons?
Iodine has a high electronegativity, which means it has a strong attraction for electrons when forming chemical bonds. It can accept electrons from other elements during chemical reactions.
6. Why is iodine stronger than chlorine?
The term “stronger” can have different meanings depending on the context. If referring to chemical reactivity, iodine is less reactive than chlorine. Chlorine is a highly reactive halogen, readily forming compounds with other elements. In contrast, iodine is less reactive, making it more stable and less likely to react with other substances.
7. Is iodine a metal?
No, iodine is not a metal. It is a non-metallic element and belongs to Group 17 (halogens) of the periodic table.
8. Can iodine dissolve in water?
Yes, iodine can dissolve in water to some extent, forming iodide ions (I^-) and triiodide ions (I3^-).
9. Is iodine reactive as a metal?
Iodine is not a metal, but it can exhibit some reactivity, particularly in its liquid state, where it forms ions and can participate in certain chemical reactions.
10. Does iodine conduct electricity at room temperature?
Solid iodine does not conduct electricity at room temperature, but liquid iodine can exhibit some electrical conductivity due to the presence of mobile ions.
11. What is the smell of iodine?
Iodine has a characteristic pungent odor that is often described as a strong, antiseptic-like smell.
12. Can iodine dissolve in alcohol?
Yes, iodine can dissolve in alcohol, such as ethanol, forming solutions with various applications in chemistry and medicine.
13. Does iodine dissolve in oil?
Iodine is not soluble in most oils but can dissolve in certain organic solvents like carbon tetrachloride or chloroform.
14. What happens when iodine mixes with metal?
The reaction between iodine and metal depends on the metal’s reactivity. Some metals, such as sodium or potassium, can react violently with iodine, producing metal iodides. Less reactive metals may form metal iodides more slowly or may not react with iodine at all.
15. Where is iodine found in nature?
Iodine is found in various natural sources, including seawater, marine organisms (e.g., seaweeds), and certain soils and rocks. It is relatively rare in the Earth’s crust but occurs in significant concentrations in the world’s oceans.
16. Is iodine an anode or cathode?
Iodine can serve as either an anode or a cathode, depending on the specific electrochemical reaction or setup in which it is involved. In electrolytic cells, it can act as an anode by releasing electrons during oxidation reactions or as a cathode by accepting electrons during reduction reactions.
17. Can iodine conduct electricity in its gaseous state?
Yes, iodine can conduct electricity in its gaseous state. When iodine is heated to high temperatures or exists in a plasma state, some of its molecules ionize, forming ions and free electrons. These mobile charge carriers enable iodine gas to conduct electricity.
18. What happens to the conductivity of iodine as it is dissolved in water?
When iodine dissolves in water, it forms iodide ions (I^-) and triiodide ions (I3^-), which increase its electrical conductivity compared to its solid form. The presence of these mobile ions allows iodine to conduct electricity to a limited extent.
19. Does temperature affect the conductivity of iodine?
Yes, temperature can affect the conductivity of iodine. As iodine is heated and changes states from solid to liquid or gas, its conductivity properties transform accordingly. Higher temperatures can lead to increased ionization and mobility of charge carriers, influencing its electrical and thermal conductivity.
20. Is iodine more or less conductive than metals?
Iodine is significantly less conductive than metals. Metals are known for their high electrical and thermal conductivity due to their delocalized electrons, which can move freely throughout the material. In contrast, iodine’s molecular structure and lack of free electrons make it a poor conductor compared to metals.
21. Can impurities affect the conductivity of iodine?
Yes, impurities can influence the conductivity of iodine. Introducing certain impurities or dopants into iodine can modify its electronic properties and affect its conductivity. For example, iodine doped with certain elements may exhibit altered conductive behavior.
22. Does the presence of moisture affect the electrical conductivity of solid iodine?
Moisture can slightly enhance the electrical conductivity of solid iodine. When iodine comes into contact with moisture, it can form small amounts of hydroiodic acid (HI), which dissociates into ions (H^+ and I^-). These ions contribute to a limited increase in electrical conductivity.
23. How does the electrical conductivity of iodine compare to other halogens?
Among the halogens (Group 17 elements), iodine has relatively higher electrical conductivity than fluorine and chlorine but lower conductivity than bromine. Fluorine and chlorine are gases at room temperature, while bromine is a liquid, which can explain their different conductive properties compared to solid iodine.
24. Can iodine exhibit superconductivity under specific conditions?
As of the current knowledge, iodine has not been found to exhibit superconductivity, which is a unique phenomenon observed in certain materials at extremely low temperatures. Superconductivity involves the complete absence of electrical resistance and is typically not observed in non-metallic elements like iodine.
25. Are there any applications where iodine’s conductivity is utilized?
While iodine is not primarily known for its conductivity, its electrical and thermal properties have some applications. For instance, iodine doped with certain compounds can be used as a semiconductor material in specific electronic devices. Additionally, its limited electrical conductivity in liquid form makes it useful in certain electrochemical reactions and as a component in some battery electrolytes.
Useful Video: Make the Iodine Clock Reaction (Chemistry)
References
- https://www.circuitsgallery.com/is-iodine-conductive/
- https://toolsweek.com/does-iodine-conduct-electricity/
- https://chemistrydictionary.org/iodine/
- https://www.chemicals.co.uk/blog/why-does-potassium-iodide-solution-conduct-electricity














Leave a Reply